Immunohistochemistry
antibody recovery and reuse in SNAP i.d.® 2.0 system
Immunohistochemistry (IHC) is a biochemical method used for the detection, localization and identification of biomarkers in tissue samples. Although IHC is used worldwide and many automated systems exist for handling large numbers of slides, research labs that manage small to medium numbers of slides at a time typically use the manual method, which can be tedious and time-consuming, and the antibody used cannot be recovered for future use.
The new SNAP i.d.® 2.0 Protein Detection System for immunohistochemistry (Merck Millipore) is a versatile, vacuum-driven system that can process from 1 to 24 slides with formalin-fixed paraffin embedded tissue (FFPE) samples as well as fresh frozen samples. The system can accommodate any typical manual protocol, and eliminates the need for a Pap pen.
For each slide, the SNAP i.d.® 2.0 system may require up to 0.6 mL of antibody solution; however, depending on the position of the tissue on the slide, this volume may be reduced. Further, the antibody can be recovered and reused several times.
In this report, we demonstrate that antibodies can be recovered and reused in the SNAP i.d.® 2.0 system several times with no signal decrease and minimal volume loss. The system was tested with FFPE samples of various human tissues. Successful IHC was demonstrated using standard monoclonal, polyclonal and prediluted monoclonal antibodies. In parallel, sections obtained from the same samples were analyzed by IHC, using a Pap pen-based manual procedure. The two procedures were performed side-by-side, applying the same reagents and incubation times to both sets of slides.
Methods and Materials
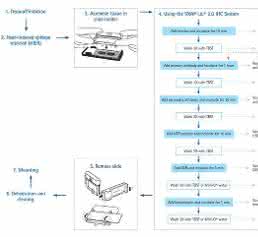
Slide deparaffinization and antigen retrieval (Figure 1, steps 1 and 2)
SNAP i.d.® 2.0 IHC method: Sections of FFPE human kidney and human and mouse brain tissue were assembled in ProbeOn™ slides (Fisher Scientific). Slides were deparaffinized using 3 xylene washes, then rehydrated in several changes of increasingly dilute reagent-grade ethanol (starting at 100 % down to 30 %), and finishing with 2 washes of Milli-Q® water (step 1). The heat-induced epitope retrieval (HIER) was performed using a Decloaker Chamber™ instrument (Biocare) for 15 min at 110 °C (step 2).
Manual method: Samples were processed manually by pipetting reagents onto slides or dipping slides into the reagents under the same incubation conditions as in the SNAP i.d.® 2.0 IHC method, but the tissue section area was outlined with a Super Pap pen (American Master Tech) prior to antibody incubation.
SNAP i.d.® 2.0 IHC system operation (Figure 1, steps 3 to 5)
Slides were assembled in the SNAP i.d.® 2.0 IHC slide holder and inserted into the IHC frame following the manufacturer’s specifications. To avoid tissue drying during the assembly of multiple slides, the slide holders were immediately filled with wash buffer (Tris-buffered saline containing 0.05 % Tween® 20 surfactant; TBS-T). Wash buffer was flushed out by initiating the vacuum and slides were then blocked for 10 min with Background Punisher™ reagent (Biocare Medical) followed by 3 TBS-T washes. Three different Merck Millipore primary antibodies were tested: rabbit polyclonal anti-aquaporin 1 diluted 1:2000, mouse monoclonal antiβ-amyloid diluted 1:500, and IHC Select® prediluted mouse anti-Alzheimer’s disease precursor protein A4 monoclonal antibody. The antibodies were incubated for 1 hour, after which they were retrieved using a pipetter and 1 mL pipette tip and kept at 4 °C until the next use. (Note: the antibodies used were kept and reused up to 5 times within a 48 day period.) Slides were washed 3 times with TBS-T followed by the addition of Mach 3™ Rabbit probe and Rabbit HRP polymer (Biocare Medical) or Mach 3™ Mouse probe and HRP polymer (Biocare Medical). Slides were incubated for 10 min each and washed as above with TBS-T.
Detection included two steps, in which slides were incubated for 3 min with Betazoid DAB chromogen kit (Biocare Medical) and then counterstained with Modified Mayer’s Hematoxylin (Sigma). Both detection steps were followed with 3 washes with Milli-Q® water. Slides were removed from the holder for the dehydration and clearing steps. Manual control slides were processed by pipetting the same reagents onto the sections for the same incubation periods as the slides in the SNAP i.d.® 2.0 system, but with smaller final antibody volumes (~ 200 µL).
Dehydration and clearing of slides (Figure 1, step 6)
Slides were dehydrated in 100 % reagent- grade ethanol (4 times, 5 min each), and cleared with xylene (3 times, 3 min each).
Mounting and observation (Figure 1, Step 7)
Slides were permanently mounted using CoverSafe™ mounting medium (American Master Tech), and analyzed on an Evos® microscope (Life Technologies), using 20-40X magnification.
Immunohistochemistry results
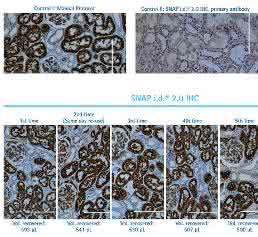
Three different antibodies (mouse monoclonal, rabbit polyclonal and mouse prediluted) were tested on kidney and brain tissues. The results demonstrate that the antibody can be successfully recovered and reused repeatedly in the SNAP i.d.® 2.0 system. Figure 2 shows a series of human kidney sections stained with rabbit anti-aquaporin 1. Aquaporins are integral membrane proteins that form water-specific channels, transporting water molecules in and out of the cell in order to increase water permeability tissues such as the kidney. The protein is clearly detected at apical and basolateral membranes of the kidney proximal tubules in the tissue sections, in both the manual and the vacuum-driven IHC experiments, even after the 5 cycles of antibody recovery and reuse. The results demonstrate that 600 µL of antibody was sufficient to stain at least 6 different slides with no loss in staining quality.
Similar results were demonstrated for amyloidβ, the major constituent of plaques in Alzheimer’s disease. Figure 3 shows five different slides of human brain cerebral cortex tissue, processed by the manual IHC protocol as well as in the vacuum-driven SNAP i.d.® 2.0 IHC system. Staining of the slides with the monoclonal anti-amyloid β revealed the typical pattern of dark spots around the neurons caused by the deposition of plaques and the amyloid β peptide.
Since it is important to recognize that several antibodies on the market are sold prediluted to a working concentration, we also challenged the SNAP i.d.® 2.0 vacuum-driven IHC system with a prediluted antibody used for the detection of the Alzheimer’s precursor protein A4 (APP). APP is widely expressed in the body but is concentrated in the synapses of neurons. Early in life, APP plays an important role in neural growth and synaptic formation and repair. Sequential proteolysis of APP is responsible for formation of amyloid β peptide. For reasons that are poorly understood, β-amyloid may accumulate to form the plaques observed in the brains of Alzheimer’s patients. In Figure 4, a series of human Alzheimer’s brain sections were stained with prediluted anti-APP antibody, and detected with anti-mouse secondary and DAB as previously described. Again, the recovery and reuse of antibodies in the SNAP i.d.® 2.0 IHC system yielded no significant difference in the staining compared to the first use in the SNAP i.d.® 2.0 IHC system or a single use in the manual IHC method.
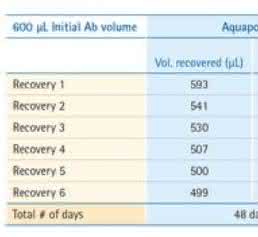
Volumetric recovery, by mass balance of the 3 antibodies analyzed, demonstrated a consistent recovery higher than 80 % even after the extended storage of 48 days at 4 °C and despite transfers from the slide holder-pipette tip and microcentrifuge tube (Table 1).
Conclusions
Manual immunohistochemistry performed with the use of a Pap pen is a common protocol for low-throughput laboratories or research facilities. While the manual method can be performed with small volumes of antibody and reagents, the process can be long, tedious, and sometimes challenging. The vacuum-driven SNAP i.d.® 2.0 IHC system offers an alternative for handling 1 to 24 slides at a time with reduced slide manipulation, easy format, no need for pap pen, reproducibility, efficient washing, and the opportunity to recover and reuse the antibodies with minimum volumetric loss and, most importantly, no decrease in sensitivity.
Author:
Sara Gutierrez
Research Scientist
E-Mail: [email protected]Merck Millipore, Corp.
17 Cherry Hill Drive
Danvers, MA 01923, 978-762-5158