Cell Growth
Consider All CO2 Incubator Parameters for Best Cell Growth
Advances in cell culture applications are occurring faster than ever before. Cultured cells are used in vaccine and pharmaceutical production, toxicity testing, regenerative medicine and cell therapy, new models in cancer, immunology, neurology and more. In these increasingly sensitive applications, the integrity and health of cells are proportionally more important.
We are gradually moving away from long established mutant cell lines and using more sensitive cells including stem cells, primary cells and 3D cultures. These cell types are better suited to mimic the in vivo environment, to give better answers to difficult biological questions, or to serve as therapeutics. Cells are constantly sampling their external milieu and reacting by turning on and off transcription, translation, cell division, etc. Thus, an understanding and awareness of how external cues affect cell growth and protein expression is paramount.
To best support these sensitive cell types and to ensure they give accurate responses predictive of the in vivo environment, new CO2 incubators designed for 21st century cell culture offer better culturing environments than previously available. In thinking about your sensitive cells, you should attend to all parameters in the incubator, because all work together to provide the complete key to cell culture success.
Temperature
Cultured mammalian cells are extremely sensitive to temperature. Just as you feel poorly when you have a fever, your cells are most sensitive to higher temperatures. While cells can survive storage at 4 °C for several days, they cannot withstand more than a few hours at more than 2 °C above normal (generally 37 °C) [1]. Look for a CO2 incubator that does not overshoot the set temperature during recovery after a door opening, and that is controlled by two temperature sensors to accurately control temperature and eliminate any chance of overheating. Often overlooked is the incubator’s recovery to set temperature. Most manufacturers will provide data showing time required to recover to set temperature after a 30 second door opening. For sensitive cell types, recovery to set temperature should be about 5 minutes following a 30 second door opening.
Gas Concentration
In the incubator atmosphere, carbon dioxide (CO2) gas is provided and works with sodium bicarbonate (or other buffer) in the cell culture medium to maintain a neutral pH of 7.0...7.4. CO2 concentration is varied depending on the composition of the selected medium, but is generally 5 %, equal to 40 mm Hg (0.05 atm) in the bloodstream, pairing with 24 mM (2 mg/mL) sodium bicarbonate. Thus, in vitro conditions created using, for example, DMEM and 5 % CO2, are a good model for the in vivo environment. Most cell culture media contain a pH indicator such as phenol red to indicate problematic changes in culture conditions. A change from pink to yellow indicates the medium is too acidic, and purple indicates too basic. Proper pH is critical for cell health, and changes from neutral can affect cell morphology and growth [2].
Some incubators provide oxygen (O2) or nitrogen (N2) gas in addition to CO2. Commonly called ”tri-gas” incubators, these provide conditions matched to specific culturing goals. O2 is provided for hyperoxic culturing applications such as research on lung or retina tissues. N2 is provided to reduce the circulating oxygen concentration to match hypoxic in vivo environments, for culturing many primary cells, stem cells, or embryos. O2 does not diffuse or dissolve into liquid nearly as fast as CO2, but it does affect nearly every cellular process including cell division and metabolism, differentiation, immune responses and more [3]. An incubator that supplies gases near the top of the humidity reservoir immediately heats and humidifies them as they enter the chamber, thus maintaining ideal conditions throughout.
Humidity
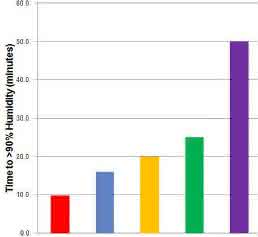
One parameter that is often overlooked in cell culture is humidity. The reason humidity is provided via a water reservoir is to limit evaporation from culture media. Evaporation of water causes concentration of trace nutrients such as salts and minerals such that they reach toxic levels. Water evaporation is four times faster at 80 % humidity than at >93 % [4]. And high humidity is the most difficult parameter to re-establish after a door opening. Different technologies offer different efficiencies of humidity recovery, as shown in Figure 2. Look for an incubator that recovers to >93 % humidity as fast as ten minutes or less after a 30 second door opening, to minimize evaporation and to ensure cells remain in carefully balanced growth medium.
Uniformity
For anyone culturing cells concurrently in many dishes/flasks, eliminating variables in conditions is critical for meaningful results that are clear and easy to interpret. Cells are affected by small differences in varied parameters and will respond. So a 21st century incubator must provide uniform temperature, gas concentration and humidity throughout, to minimize variation in cell responses that are reflected in confusing experimental results. This is why uniformity from top to bottom and side to side in all areas of the incubation chamber is critical.
Recovery
Recovery to set parameters after every door opening is a key component of a 21st century incubator. When considering a new incubator, look for recovery data not just for temperature or CO2 but also for humidity, since all work together to provide the optimum environment. Part of delivering fast recovery is placing all sensors inside the chamber, to experience and react to the same conditions as your cells. Some incubators place their sensors in a “by-pass loop”, requiring a sample of air to be extracted from the chamber, passed through tubing and a pump and over the sensor. This external equipment requires a separate heater as well. The air is then put back into the chamber. A by-pass loop carries the risk of contaminants lodging in the external parts and means the sensors are experiencing different conditions than your cells, resulting in delayed and less than ideal reactions from the sensors.
Air Circulation
A circulating fan in the incubation chamber provides uniform conditions and maximum recovery speeds. There are some stories about why a fan is a bad idea in a CO2 incubator, but these are not accurate for all incubators with a circulating fan. Only an incubator with an in-chamber fan can provide maximum uniformity and minimal recovery times. This is why the European Pharmacopoeia requires forced air circulation for dry heat sterilization [5]. Look for an incubator that provides gentle circulation. The fan should not blow directly on the cultures but instead should direct air around the sides. This turbulence means the fan will not dry out cultures. A fan based on a DC motor means no vibration. In fact, in many labs, vibration in cultures is actually due to external forces including equipment such as a centrifuge or ultra low freezer, or building construction. A fan located in the external jacket does not provide fast recovery to the inner chamber, which is separate from the jacket.
Contamination Control
Humans carry 10,000 micro-organisms/cm2 on skin surfaces [6]. And indoor room air carries from 30 to 700 micro-organisms/m3 [6]. Clearly, micro-organisms are our constant companions.
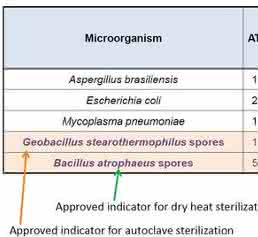
An on-demand, automated sterilization cycle is a wonderful feature for a 21st century CO2 incubator, and different models offer varied versions and options. Understanding and comparing the differences can be confusing. Because of this, the European Pharmacopoeia (EP) and the United States Pharmacopeia (USP) now require proof of dry heat sterilization using an “overkill” approach and an approved biological indicator organism, Bacillus atrophaeus [5] (a.k.a. Bacillus subtilis [8]), a heat and dessication resistant endospore forming bacterium. Both the EP and the USP require proof of elimination of at least one million of these resistant bacteria, resulting in a less than one in one million chance that any life survived, for a Sterility Assurance Level (SAL) of 10-6. The USP further requires an overkill approach to result in a 12-log reduction. Here, a 6-log reduction may be demonstrated using half the lethality, for example, sterilization at a given temperature for half the standard time.
The Thermo Scientific Steri-Run™ high temperature sterilization cycle has been independently tested [9] and proven to eliminate >6 logs of resistant bacterial spores in 45 min. at 180 °C. This is half the normal 90 min. cycle, proving a total 12 log reduction that complies with EP and USP requirements to prove an overkill sterilization using dry heat. The results show elimination of all A. brasiliensis (a black mold commonly found in older buildings), E. coli (a common research laboratory bacterium), M. pneumonia (one of many species of mycoplasma, a common cell culture contaminant), B. atrophaeus (spore form used for testing dry heat sterilization), and G. stearothermophilus (extremely resistant spore form used for testing autoclave sterilization). Four locations in the chamber were each tested three separate times, and the results averaged (Figure 3).
But an on-demand cycle is not enough. These are ideal for eliminating resident micro-organisms and can save time by simplifying cleaning and eliminating the need to remove and autoclave parts. However, as noted above, micro-organisms are constantly circulating in the air and shedding from our skin. Every time you open the incubator door, micro-organisms can enter. Regardless of whether there is active air circulation from a fan, conditions inside the incubator are so different from the ambient room conditions that the air will rush to equilibrate. And small air currents created from opening the door can sweep dirt and contaminants inside. So a 21st century incubator should also offer an all-the-time contamination prevention method that will keep incoming bacteria and fungi at bay.
There are only a few options for CO2 incubators that offer constant contamination prevention. One is a HEPA filter, requiring a fan to circulate the air over the filter. An H13 HEPA filter with active air circulation can provide ISO 5/Class 100 cleanroom conditions within 5 minutes after every 30 second door opening [10], but the speed to ISO 5 conditions depends upon the design of the airflow, so look for independent test results for different incubators. ISO 5/Class 100 conditions means the air in the chamber is 4 logs or 10,000 times cleaner than normal indoor room air [11]. A common misconception about HEPA filtration is that it is limited to capturing particles 0.3 µm or larger. This comes from the fact that due to the physical processes involved, 0.3 µm particles are the most difficult size to catch. This is why it is called the Most Penetrating Particle Size (MPPS). H13 HEPA filters are rated at an efficiency of 99.97 % for 0.3 µm particles, and larger and smaller particles are caught with even greater efficiency [12]. And research indicates that micro-organisms do not survive or escape from HEPA filters [12]. You should dispose of them in your standard laboratory biological waste containers which are autoclaved prior to disposal.
Naturally beneficial metal surfaces such as solid copper are well known to be effective in controlling micro-organisms, and copper is the world’s most recycled material [13]. Pure metal content, and higher temperature and humidity augment the effects [14]. Stainless steel is not inherently antagonistic to micro-organisms [14] but electropolishing stainless steel removes microscopic structure that can help hide micro-organisms. Thus, there are good reasons to consider a CO2 incubator with one of these optional interiors.
Ultraviolet (UV) light is used in many applications to control microorganisms, but unfortunately it is not a good choice for CO2 incubators, due to its ineffectiveness in humidity higher than 70 % [15] – most CO2 incubators generate humidity at 80 % or greater. In addition, UV light bulbs lose intensity over time and areas in shadow are not disinfected, leaving survivors to propagate.
Conclusions
For today’s specialized cultured cells and applications, a CO2 incubator incorporating 21st century technologies is required. Such an incubator is validated for all operating parameters and offers data that you can carefully evaluate. Enough information should be provided that you can understand what is presented and consider in the same way that you would any other experimental results. Look for proof of uniformity of conditions throughout the chamber, provided by active airflow circulation. Temperature, gas concentration and humidity all work together to provide conditions critical for your cell culture success, and the time your cells spend at optimum conditions will be reflected in your downstream processes and data. Fast recovery to set conditions after every door opening will ensure cells grow at their prescribed conditions, resulting in clear results for your research and production goals. In-chamber sensors will constantly monitor and control the conditions surrounding your cells, which a by-pass sensor cannot do. And since micro-organisms are our constant companions, ensure that your incubator choice offers not only an independently proven sterilization that eliminates bacterial spores, but also an effective, constant, contamination prevention method.
References
[1] Freshney RI. Culture of animal cells: a manual of basic technique and specialized applications, 6th ed. John Wiley & Sons, Inc., Hoboken NJ, United States. 2010.
[2] Gibco. Handbook for cell culture basics. Invitrogen Life Technologies B-087243 0110.
[3] Bates MK. Culturing cells under hypoxic conditions for biologically relevant results. Am Lab 2012.
[4] Esser P and Weitzmann L. Evaporation from cell culture plates. Thermo Fisher Scientific TILSPNUNCBU02 0111, 2011.
[5] European Directorate for the Quality of Medicines & HealthCare, Council of Europe. European Pharmacopoeia, 8th ed. Strasbourg France, 2014.
[6] Grice EA et al. A diversity profile of the human skin microbiota. Genome Res. 18, 2008.
[7] Stryjakowska-Sekulska M et al. Microbiological quality of indoor air in university rooms. Polish J Environ Stud 16(4), 2007.
[8] The United States Pharmacopeial Convention. The United States Pharmacopeia, 37th Ed. Rockville MD, United States. 2014.
[9] Public Health England. An evaluation of the effectiveness of the sterilization cycle of the Thermo Scientific™ CO2 incubator with the Thermo Scientific Steri-Run™ cycle. Report No. 14/036, 2014.
[10] Importance of Class 100 air in a CO2 incubator. Thermo Fisher Scientific, WPCO2Class100 1109, 2009.
[11] Enterprise and Industry Directorate-General, European Commission. EudraLex: The rules governing medicinal products in the European Union. Volume 4: EU guidelines to good manufacturing practice, medicinal products for human and veterinary use. Annex 1: Manufacture of sterile medicinal products, 2008.
[12] National Institute for Occupational Safety and Health, Centers for Disease Control and Prevention, Department of Health and Human Services. Guidance for filtration and air-cleaning systems to protect building environments from airborne chemical, biological, or radiological attacks. 2003.
[13] Jolly J. The U.S. copper-based scrap industry and its by-products. 12th ed. Copper Development Association Inc. New York, NY, United States, 2012.
[14] Grass G et al. Metallic copper as an antimicrobial surface. Appl Environ Microbiol 77(5), 2011.
[15] Burgener J. Position paper on the use of ultraviolet lights in biological safety cabinets. Appl Biosafety 11(4), 2006.
Mary Kay Bates, M.S.
Global Cell Culture Specialist, Thermo Fisher Scientific, [email protected]